Liposomal herbal preparations

TABLE OF CONTENTS:
Herbal Preparations
Today, scientists have succeeded in confirming the health-promoting activity of many chemical compounds present in plants. Their effects on cells may be certain, but an ongoing problem with phytotherapy is the bioavailability of the beneficial ingredients. Currently, many researchers are focusing on finding the most favorable routes of administration for active compounds, increasing absorption efficiency, improving bioavailability, and formulation technologies for supplements and herbal medicines. One extremely favorable formulation in terms of bioavailability is liposome-based products1.
The most appropriate form of taking herbal supplementation is oral. After ingestion, food and biologically active ingredients are digested and then absorbed in the digestive tract. Unfortunately, when thinking about supplementation and phytotherapy, we must be aware that a large proportion of essential molecules are poorly absorbed. Many oral medications and herbal compounds, have very low levels of absorption. For some substances, less than 10% of the molecules are absorbed into the bloodstream1.
Absorption of active molecules into the bloodstream depends on their solubility in water, their lipophilicity (ability to dissolve in fats), as well as their rate of penetration through cell membranes, stability against various enzymes (sensitivity to the body’s metabolic processes) and resistance to acids and bases. By protecting molecules from destruction and increasing their ability to pass through cell membranes, liposomes increase the bioavailability of plant-based supplementation1,2.
What are liposomes?
Liposomes are spherical forms in which bioactive substances can be encapsulated inside. They are most often formed from phospholipid molecules (components of the membranes of the cells that build our bodies). Liposomes have been successfully used to alter the pharmacokinetic profile of drugs, as well as herbs and vitamins. Due to their unique properties, liposomes are capable of enhancing the potency of plant substances by increasing their solubility, improving their bioavailability and increasing their stability in the human body2.
In their interior, liposomes encapsulate a fraction of the solvent in which active substances can be found. They can have one, several or many spherically closed internal membranes. Liposomes are made up of polar lipids, which are characterized by having a lipophilic group and a hydrophilic group. This means that their one pole adheres to water-soluble substances and the other to fat-soluble substances. When exposed to water, polar lipids self-organize and form vesicular forms2,3.
How do liposomes increase the bioavailability of active ingredients?
A liposome has a core of aqueous solution surrounded by a hydrophobic membrane in the form of a lipid bilayer. Hydrophilic substances dissolved in the core cannot easily pass through the bilayer, so they are protected from external agents. Hydrophobic (non-water-soluble, fatty) chemicals bind to the lipid bilayer. Thus, they are dispersed in the water environment and are more easily absorbed by the body. This means that the liposomal form of the herbal preparation increases the bioavailability of both fat-soluble and water-soluble ingredients. As active molecules are delivered to the site of action, the lipid bilayer of the liposome fuses with other bilayers, such as the cell membrane, thereby delivering the contents of the vesicle1-3.
In summary, the beneficial effect of liposomes on nutrient bioavailability is due to several of their pathways of action. It is:
- An increase in the solubilization of the active substance;
- Protection of active ingredients from degradation;
- Increasing the absorption of the active substance into the bloodstream.
Liposomes in nature
A great example of a natural nutritional fluid containing liposomes is human milk. Due to its unique properties, this secretion has been studied in depth by scientists for decades. As a result of this scientific work, the components of breast milk and their relationship to infant health and development are very well understood. Analyses of the chemical composition of human milk have shown that it is an extremely complex suspension containing more than 200 fat- and water-soluble components. The dominant forms in milk are emulsion droplets and casein micelles. Recently, electron microscope studies have also detected lipid vesicles (liposomes)1 in it.
Use of liposomes in the manufacture of drugs and supplements
Until recently, the use of liposomes in medicine mainly concerned targeted drug delivery. This term is used to describe preparations designed to selectively act on certain types of cells, such as cancer cells. In recent years, there has been a clear trend of using the properties of liposomes to increase the effectiveness of dietary supplements and herbal preparations. This new approach stems from the problem of low absorption and bioavailability rates of traditional oral diet pills and capsules. Unfortunately, the low bioavailability of supplements taken orally is an issue well documented in clinical trial results. Therefore, the natural “encapsulation”, on a microscopic scale, of lipophilic and hydrophilic nutrients in liposomes is an effective method of protection against destructive enzymes and acids secreted by the digestive system. In addition, the encapsulation of the substance in liposomes increases the efficiency of its delivery to cells and tissues4.
Liposomes unequal liposomes – what affects the quality of liposome-based formulations?
It should also be noted that there are certain factors that affect the percentage of liposomes in the formulation, their stability and the actual amount of realized entrapment of the active substance in them. They are:
- A method for producing and preparing liposomes;
- The composition, quality and type of phospholipid used to formulate and manufacture liposomes;
- the content in the formulation of uniformly sized liposome vesicles, which are stable and maintain their closed charge.
The aforementioned elements form the basis for developing effective liposomal carriers for use in the production of supplements and specialty foods.
The most important types of liposomes include multilayer vesicles (MLV), small monolayer vesicles (SUV, with one lipid bilayer) and large monolayer vesicles (LUV). For drugs and supplements, multilamellar liposomes are the least desirable form. This is because MLVs are large vesicles with more smaller vesicles in them, making it difficult to access the active ingredient.
Among liposomes found in orally administered formulations, monolayer vesicles are the most favorable form. Prominent among them are LUV liposomes having a diameter of more than 50 nm. They exhibit a longer storage life than small SUV vesicles because they do not experience high stresses at the level of the double lipid layer due to their larger size. LUVs can be successfully used as carriers of active substances due to their optimal ratio of aqueous to lipid phase. In addition, the lipid membrane of LUV vesicles shows great similarity to the cell membrane of human cells3.
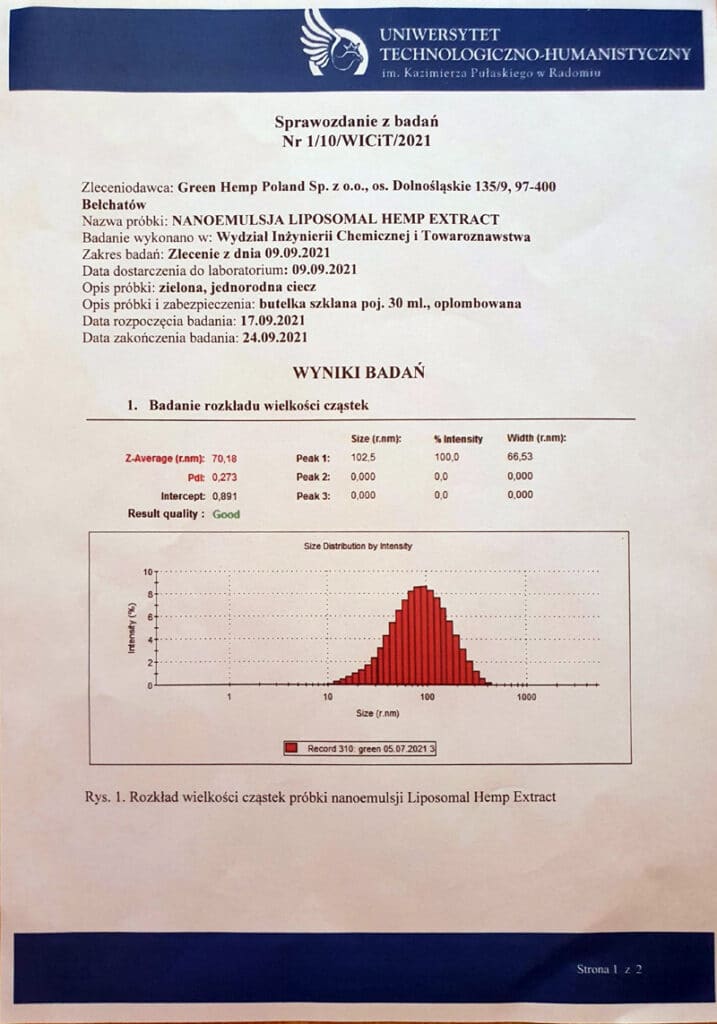
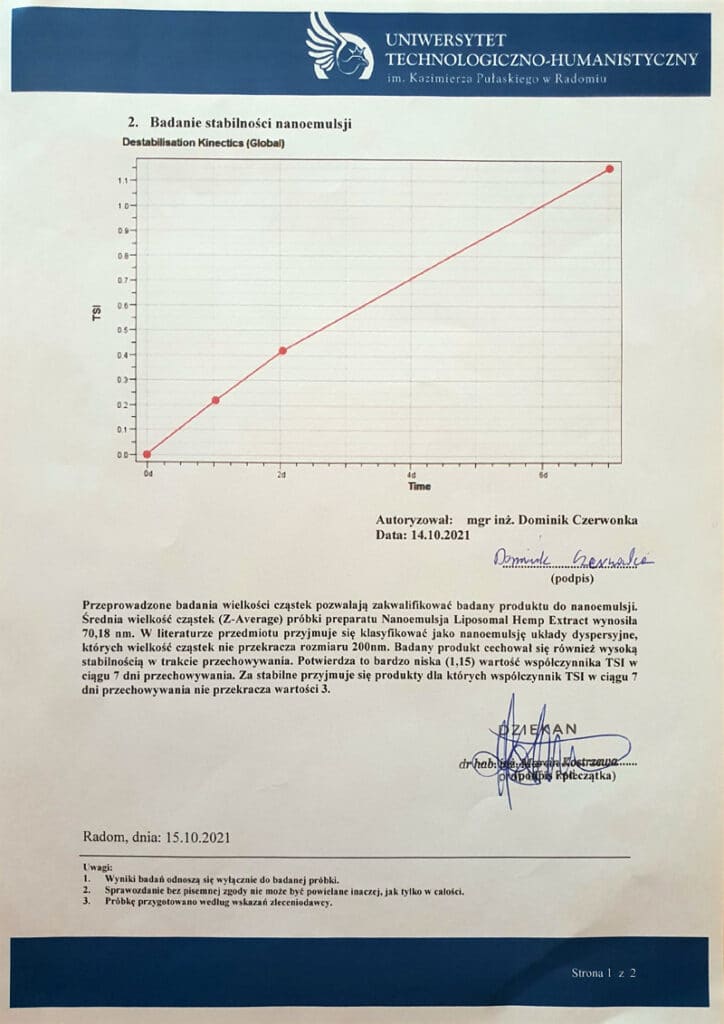
In tests carried out at the laboratory of the University of Technology and Humanities named after him. Kazimierz Pulaski in Radom showed that Nanoemulsion Liposomal Hemp Extractcharacterized an average bubble size of 70.18 nm. This means that it falls into the category of LUV liposome-based formulations, with the associated favorable increase in the bioavailability of both lipophilic and hydrophilic components. In addition, the laboratory confirmed the high stability of the nanoemulsions, which is one of the most important quality parameters for liposome-based formulations. At the same time, the vesicles in the formulation were shown to be small enough to qualify as nanoemulsions. This term describes a system in which the size of the dispersed particles does not exceed 200nm. Nanoemulsions are characterized by the ease of introduction of biologically active compounds, both into the internal phase and into the dispersing phase, which positively influences their bioavailability5.
Content and development
Dr. of pharmacy Sara Janowska
References
- Keller, B. C. Liposomes in nutrition. Trends in Food Science & Technology 12, 25–31 (2001).
- Porter, C. J. H., Trevaskis, N. L. & Charman, W. N. Lipids and lipid-based formulations: optimizing the oral delivery of lipophilic drugs. Nature Reviews Drug Discovery 2007 6:3 6, 231–248 (2007).
- Technologia małych cząstek – liposomy, mikrocząstki, mikrokapsułki, mikrosfery, nanocząstki lipidowe. – Aptekarz Polski. https://www.aptekarzpolski.pl/wiedza/technologia-malych-czastek-liposomy-mikroczastki-mikrokapsulki-mikrosfery-nanoczastki-lipidowe/.
- DeFelice, S. L. The nutraceutical revolution: its impact on food industry R&D. Trends in Food Science & Technology 6, 59–61 (1995).
- Idzikowska, M. et al. NANOTECHNOLOGIA W PRODUKCJI ŻYWNOŚCI-KIERUNKI ROZWOJU, ZAGROŻENIA I REGULACJE PRAWNE. (2012).
Customer review
In mid-February 2022, I got severe pain in my lumbar spine, which radiated to my lower limbs.
After a visit to a neurologist, I had an MRI scan of this section of the spine, on which, in addition to herniated discs, it revealed in the spinal canal at L1, L2, an otorrhized dense fluid space 10mm wide and 23mm long, compressing the contents of the meningeal sac, with suspected meningioma. (MRI opia attached).
After consultation, the neurosurgeon recommended an MRI with contrast, which I had performed in the second half of March. This examination revealed the presence of a 6x15x23 mm pathological mass at the level described above, suggesting the presence of meningioma-like lesions. (description attached)/
The neurosurgeon, after seeing the result of the above test, recommended surgery. He concluded that the meningioma is not reabsorbing, although it is not causing pressure at the moment, and surgery can be withheld while observing further development.
He recommended a follow-up examination after 3 months.
After the visit, I contacted a representative of green hemp sp. Ltd. who prepared me“Liposomal Herbal Preparations” products, which were supposed to cause this pathological mass to be absorbed.
These products, I took for almost 3 months, until the follow-up MRI on 23-06-22.
As you can see from the attached test description, the meningioma, diagnosed earlier, has completely absorbed.
Many thanks to Mr. Hubert of Green Hemp for preparing an effective therapy.
I heartily recommend this company’s products to others in need!
I want to add that during the observation period (from the 2nd examination to the follow-up examination) , in addition to the above, I did not take any other agents.
Wieslaw Kaliski